A panel of FDA experts recommended that Americans be assisted in combating opioid overdoses. The opioid crisis is still raging across the country.
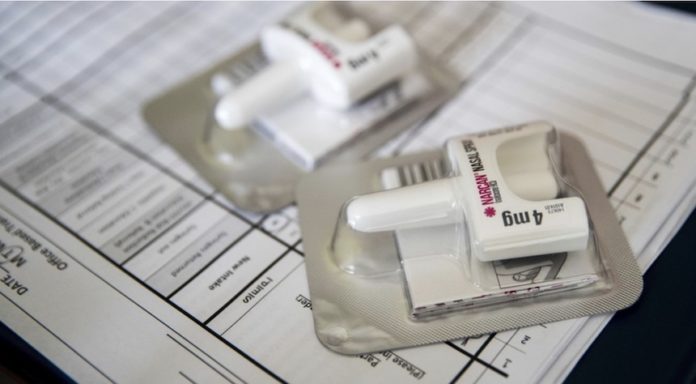
The panel recommended this week that the overdose-reversing drug Naloxone be made over the counter. Although it is most commonly known as Narcan, the drug can also be called by other names.
As it stands now, Narcan is available in all 50 states’ pharmacies. Experts warn that although pharmacists are allowed to sell Narcan to anyone who asks them, the instructions for Narcan use can sometimes be confusing. Due to the stigma associated with drug use, some people might not wish to visit a pharmacy to ask for Narcan.
Narcan is available anonymously in drugstores, grocery stores, and vending machines.
Dr. Jody Green of the FDA stated, “We believe nonprescription Naloxone could help to address these barriers. ”
The panel demanded that Emergent Biosolutions (Narcan’s manufacturer) revise the instructions. Emergent Biosolutions stated that they would address packaging issues but insists that focus group users were able to navigate the instructions.
According to the Associated Press Emergent said that it would move all directions towards a single panel and add pictograms as per FDA’s request.
The panel recommends Emergent immediately address the issue with instructions.
Maria Coyle, chair of the panel and Ohio State University pharmacist professor, stated that the possibility of delay in the availability of the product is greater due to the current crisis and its devastating consequences.
The panel’s vote was non-binding, but it was unanimous among its members
The FDA will take the recommendations of the panel into account, but community advocates can take heart in the vote. Narcan stigma can be removed and Narcan can be made more accessible.
Sheila Vakharia, deputy director of academic engagement, at Drug Policy Alliance, told the AP that the deputy director of drug policy will have a significant effect on how people view the drug.
It will amaze me to see what FDA does next.